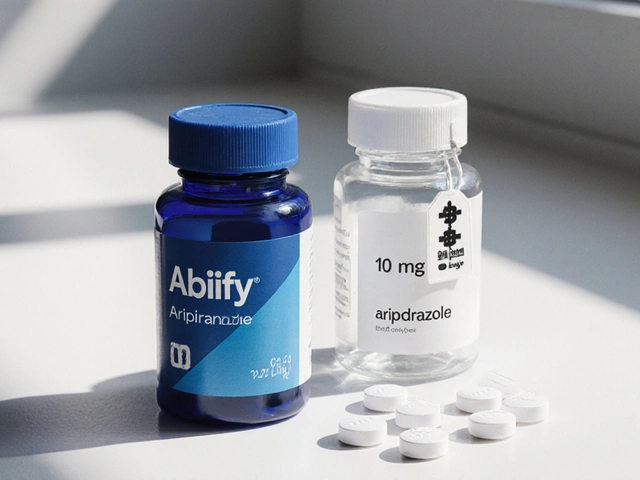
Have you ever stared at your prescription bottle, confused by the small print under the bold name you were expecting? You ask for the medication by its recognizable name, but the bottle arrives with something else written on it. It happens all the time. This mix-up isn't just annoying-it can make people wonder if they got the wrong medicine. Understanding the distinction between generic and brand names on Rx labels is the first step toward taking control of your health and your wallet.
This isn't about complicated chemistry lessons. It's about knowing exactly what's inside the pill you swallow and why the price tag changes so drastically. Let's look at the details on the label, what the rules actually say, and how you can verify you're getting the right care without paying extra.
What Exactly Are Generic and Brand Names
When you pick up a script, you are dealing with two naming systems running side by side. One is the proprietary identity created by the company that invented the drug, and the other is the chemical reality of what is actually working in your body.
Brand-name drugs are proprietary trademarks created by pharmaceutical manufacturers that identify a specific product made by a specific company. Think of this as the "logo" of the medicine. When a company develops a new treatment, they spend years proving it works. They earn the right to sell it under a unique name they chose. For example, Prilosec was a brand-name drug for reducing stomach acid.
Then there is the other side of the coin.
Generic drugs are medicines that contain the exact same active ingredient as the brand-name version but use the chemical name instead of the trademark. Using the earlier example, omeprazole is the generic name for Prilosec. It has the same job, the same strength, and works in the same way.
The physical appearance often differs. The pill might be a different color, shape, or size. Manufacturers cannot copy the original design due to trademark laws. This leads to confusion, but the therapeutic effect remains the same.
The Money Difference: Why Generics Cost Less
If they do the same thing, why does the bill jump from $400 to $4? This question sits at the heart of modern healthcare economics. The gap exists because of who pays for the initial risk.
Brand-name companies carry massive upfront costs. Before a single patient takes a brand pill, that company funds clinical trials, patent filings, and marketing. One analysis suggested these costs average around $2.6 billion for an approved drug. That investment needs recouping through higher prices during the patent period.
Generic manufacturers do not repeat that expensive trial process. They simply prove their version works just as well. Because they skip the multi-billion dollar R&D phase, they can offer the medicine much cheaper. In recent years, generics have saved the U.S. healthcare system approximately $1.67 trillion over a decade. On an individual level, savings typically range between 80% and 85% compared to the brand equivalent.
| Factor | Brand-Name Drug | Generic Drug |
|---|---|---|
| Active Ingredient | Identical | Identical |
| Research Costs | High ($2.6B average) | Low (No new trials) |
| Patient Cost | Full Price | Typically 80-85% Less |
| Appearance | Trademarked Color/Shape | Different Color/Shape |
Consider atorvastatin. The brand name Lipitor used to cost nearly $300 a month for patients. Once the generic entered the market, that same cholesterol-lowering medicine dropped to around $4. That difference allows millions of people to afford life-sustaining treatments that were previously out of reach.

Safety Standards and Bioequivalence
Is it safe? This is the big concern. Many people worry that "cheap" means "unsafe." The regulatory bar is high enough that this fear is largely unfounded in developed systems.
The Food and Drug Administration (FDA) set strict rules for this via the Hatch-Waxman Act. This legislation created a pathway that lets generics be approved faster, provided they meet rigorous standards. They aren't just guessing; they have to prove mathematically that the drug behaves the same way in the bloodstream.
Bioequivalence is a requirement showing that a generic drug delivers the same amount of active ingredient into the blood at the same speed as the brand-name drug. Specifically, the confidence interval for absorption measurements must fall within 80% to 125%. This margin is clinically insignificant for 99% of drugs.This means the generic contains the identical active ingredient in identical strength. A study from UCSF noted that "generics contain the same active ingredient as the branded version, be the same strength and format, and work the same way in the body." The quality and manufacturing standards match exactly.
However, there are nuances. Some medications have a narrow therapeutic index. These are drugs where tiny changes in dosage could affect outcomes, such as warfarin. For these rare cases, doctors might insist on sticking to the specific manufacturer to ensure consistency, though even then, the regulations remain tight. For the vast majority of prescriptions-from antibiotics to antidepressants-the interchangeability is seamless.
How to Read Your Prescription Label
Labels are designed to tell you everything you need to know, but the text can be dense. Knowing where to look saves you from confusion.
- Check the Top Line: The largest text usually displays the brand name if it exists, or the generic name. Often, both appear.
- Look Beneath the Header: The chemical name (the generic identifier) often appears in smaller font below the prominent text. It tells you exactly what molecule is inside.
- Verify the Strength: Numbers like "5 mg" or "100 mg" confirm the potency matches your doctor's order.
- Note the Manufacturer: Sometimes listed near the bottom, this shows which pharmacy supplier provided the stock.
Most states require pharmacists to include both names. About 90% of prescriptions filled now are for generic drugs. Seeing the generic name shouldn't signal alarm; it signals value. If you prefer a brand name, you have the right to request it. Simply tell your doctor to mark the prescription "dispense as written," though insurance may charge you the full price difference.

Patient Concerns and Real World Experience
Data suggests satisfaction is high once the education gap closes. University Hospitals reported that 78% of patients feel satisfied with generics after receiving a brief explanation. Yet, confusion persists. A CVS Health survey found that 24% of patients felt confused by the different-looking pills.
Why do some people report feeling different effects? Occasionally, inactive ingredients-fillers, binders, or dyes-vary between brands. While these don't change the active medical effect, some individuals have sensitivities to specific fillers. If you notice unusual side effects, talk to your pharmacist about the inactive ingredients rather than assuming the medicine isn't working.
There are also logistical advantages to the generic system. With 15% of medication errors involving confusion between similar-looking pills, distinct packaging helps. Even though generics can't look like the brand, having them look distinct prevents mixing up bottles accidentally.
Future Trends in Drug Naming and Availability
The landscape is shifting again with biologics. Unlike simple chemical pills, biologic drugs are complex molecules grown in cells. When patents expire, we see "biosimilars" rather than traditional generics. These are priced lower (15-30%) but not as dramatically cheap as standard generics. As more biologics lose patent protection by 2026, you will see these names appear more frequently on labels.
Supply chain resilience is another factor. Global production relies heavily on specific regions for raw materials. Regulations like GDUFA III aim to speed up approvals to ensure supply stability. Regardless of trends, the core promise remains: the active medicine on your label must be effective and safe.





Molly O'Donnell
April 2, 2026 AT 10:36Ive seen plenty of people panic over that label text but theyre usually wasting their time on it.
Rod Farren
April 2, 2026 AT 12:18The regulatory framework governing these distinctions is actually quite rigorous when you examine the clinical data. Most folks worry about the excipients rather than the API which is statistically insignificant for most therapies. Bioavailability metrics are standardized to ensure the confidence interval remains within the acceptable 80-125 percent window. You really need to understand that the active pharmaceutical ingredient is identical molecule for molecule. Manufacturers bypass the preclinical phases entirely which drives down the capital expenditure significantly. That cost reduction trickles down to the patient co-pay structures we see today. However narrow therapeutic index medications require closer monitoring by the prescriber regardless of formulation. The FDA oversight mechanisms are designed specifically to prevent adverse events due to switching. Pharmacists often handle the dispensing decisions without even consulting the physician on interchangeability. If there is a concern regarding the fillers then a dialogue about allergic reactions should happen immediately. It is crucial to remember that the patent cliff allows for market competition to stabilize pricing. Generic substitution laws vary by jurisdiction but the federal standard remains consistent in quality control. Reading the label closely helps avoid confusion with look-alike sound-alike medication errors. Insurance formularies drive the majority of these switches rather than patient preference alone. Ultimately the health outcome should remain the primary metric of success for any therapeutic intervention.
Owen Barnes
April 3, 2026 AT 12:59The distinction between excipents and API is reallly critical.
We must ensure saftey standars remain high for evryone.
Callie Bartley
April 3, 2026 AT 13:28It makes me sad that we dont trust our own domestic pharma anymore.
The whole system feels rigged against the average citizen paying full price.
Cara Duncan
April 5, 2026 AT 11:33I get why you feel that way but prices are better now :)
We should focus on what works for our bodies 💊
Russel Sarong
April 6, 2026 AT 01:18OH! MY! GOD!!! The savings potential is absolutely massive!!!
I cannot believe how much money families used to lose on brand names!!!!
This information is a life saver for everyone dealing with chronic conditions!!!!!
Arun Kumar
April 6, 2026 AT 06:18Healthcare access is a global issue we must solve together.
Understanding the difference empowers patients from every background.
Let us support each other in making informed choices.
James DeZego
April 7, 2026 AT 04:26Exactly right about empowerment being key :)
Knowledge reduces anxiety significantly.
Keep spreading the word friends !)
Rocky Pabillore
April 8, 2026 AT 13:44While sentiment is nice it lacks nuance.
The layman rarely understands the supply chain complexities.
But perhaps they do not need to anyway.