Every time you fill a prescription, you might be handed a pill that looks different from the one you’ve taken before. Maybe it’s a different color, shape, or has a strange logo on it. You might wonder: Is this just a copy? Is it safe? Will it even work the same way? These questions are common-and they’re based on myths that have stuck around for decades.
Generic drugs aren’t knockoffs. They’re legally required to be the same.
Let’s clear this up right away: generic drugs are not fake, inferior, or watered-down versions of brand-name medications. They are exact copies in every way that matters-medically. The active ingredient, the strength, how it’s taken (pill, injection, liquid), and how your body absorbs it are all identical to the brand-name version. The U.S. Food and Drug Administration (FDA) doesn’t just say this-they prove it.
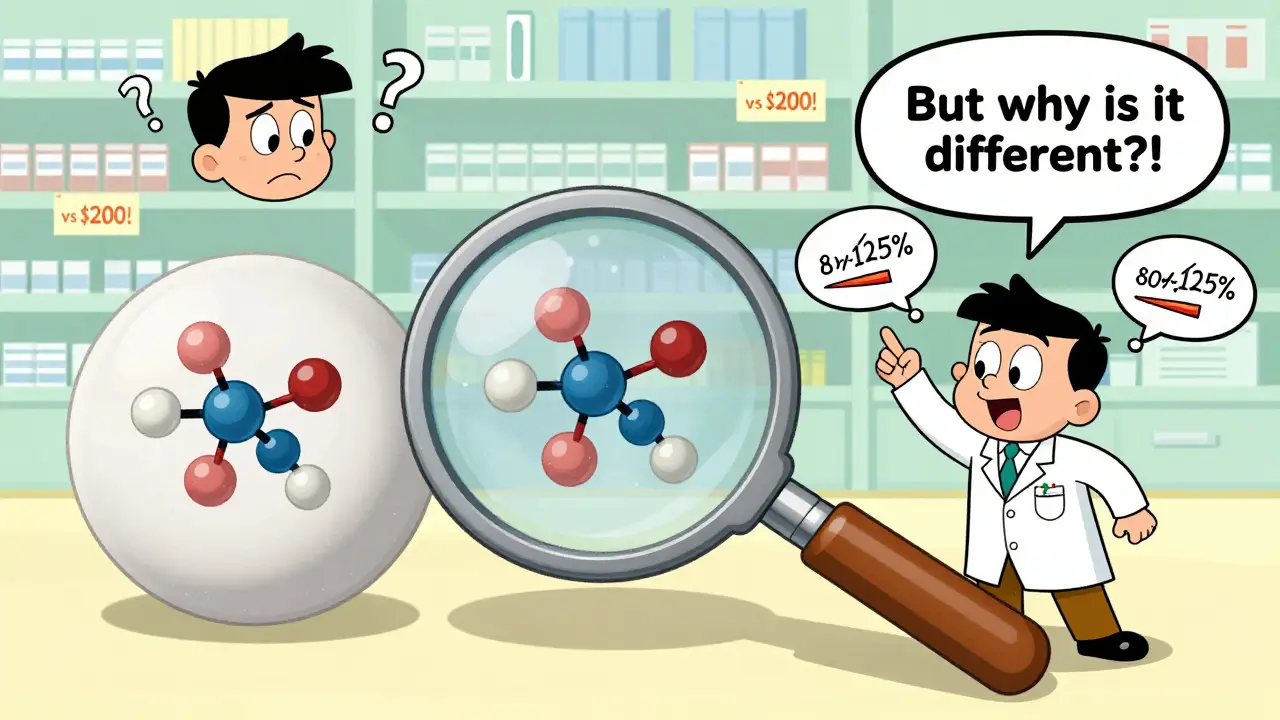
Before a generic drug hits the shelf, the manufacturer must submit an Abbreviated New Drug Application (ANDA). This isn’t a shortcut-it’s a rigorous process. The FDA requires proof that the generic delivers the same amount of active drug into your bloodstream at the same rate as the original. That’s called bioequivalence. The standard? The 90% confidence interval for absorption must fall between 80% and 125% of the brand-name drug. In plain terms: your body processes it the same way. No guesswork. No loopholes.
And it works. In 2022, 90% of all prescriptions filled in the U.S. were for generic drugs. That’s not because people are settling. It’s because they work.
So why do they look different? And why are they cheaper?
Here’s where the confusion starts. Generic drugs look different because the law says they have to. Trademark rules prevent generics from copying the exact shape, color, or logo of the brand-name drug. That’s why your levothyroxine might be white today and pale yellow next month. The active ingredient? Still 100% the same.
The price difference? That’s where the real story lies. Brand-name drugs are expensive because the company spent years and hundreds of millions developing the drug, running clinical trials, and marketing it. Once the patent expires (usually after 20 years), other companies can make the same drug without repeating those costs. They don’t need to run new safety trials because the original drug’s safety has already been proven.
That’s why a 30-day supply of generic atorvastatin (Lipitor) costs around $4, while the brand version can run over $200. The Congressional Budget Office found generics cost, on average, 85% less. That’s not a trick. That’s how competition works.
What about those side effects? I felt different after switching.
Some people swear they feel different on a generic. Maybe they got a headache. Maybe they felt more tired. Or maybe their stomach felt off. In most cases, it’s not the active ingredient. It’s the inactive ones.
Generics can use different fillers, dyes, preservatives, or binders. These don’t treat your condition-they just hold the pill together or make it easier to swallow. But some people are sensitive to them. Lactose, for example, is a common filler. If you’re lactose intolerant and your new generic has it, you might feel bloated. That’s not the drug failing. That’s your body reacting to something in the shell.
Pharmacists report spending an average of 3.2 minutes per prescription explaining this. Most patients don’t realize that the “side effects” they’re feeling aren’t from the medicine itself, but from something added to make the pill look or taste different.
Are there exceptions? Yes-but they’re rare.
There’s a small group of drugs where even tiny changes in how the body absorbs the drug can matter. These are called narrow therapeutic index (NTI) drugs. A little too much? Toxic. A little too little? Ineffective.
Examples include:
- Warfarin (blood thinner)
- Levothyroxine (thyroid hormone)
- Phenytoin and carbamazepine (anti-seizure meds)
For these, doctors sometimes prefer to keep patients on the same brand to avoid any variability. But even here, studies show that 92% of patients switch successfully without issues-especially when monitored closely. The Epilepsy Foundation found that 17% of patients reported breakthrough seizures after switching generics. But follow-up analysis by the FDA showed most of those cases weren’t due to bioequivalence failure. They were linked to other factors: missed doses, stress, illness, or even changes in diet.
The bottom line? For 96% of all medications, switching to a generic is as safe and effective as staying with the brand. The FDA reviewed over 1,800 reports of possible generic-related issues between 2018 and 2022. Only 5.5 cases per year were confirmed as actual bioequivalence failures.
How do we know generics are really the same?
The FDA doesn’t just trust manufacturers. They test them. Every batch. Every year. They inspect manufacturing sites, check for contamination, and verify that the active ingredient is present in the exact amount claimed. In a 2022 analysis of 2,070 generic drugs, the FDA found the average active ingredient content was 99.2% of the labeled amount. That’s tighter than the tolerance for most food products.
And it’s not just the U.S. The European Medicines Agency, Health Canada, and Australia’s TGA all have the same standards. If a generic drug is approved in one of these countries, it’s been held to the same rigorous standards.
Real-world data backs this up. A Kaiser Family Foundation survey in 2022 found that 89% of people who got generics were satisfied with them. Medicare Part D beneficiaries saved over $500 a year on average just by switching. And on Drugs.com, 82% of users reported generics worked just as well as brand-name drugs.

Can pharmacists switch my prescription without asking?
In 49 out of 50 U.S. states, yes. Pharmacists are allowed to substitute a generic for a brand-name drug unless the doctor writes “Dispense as Written” or “Do Not Substitute.” Mississippi is the only state that requires a doctor’s note for substitution.
But here’s the twist: if you prefer the brand-name drug, you can still ask for it. In 28 states, pharmacies are required to give you the brand-name version at the generic price if it’s therapeutically equivalent. That’s not a loophole-it’s consumer protection.
Still, 65% of patients ask for brand-name drugs out of fear, even when their doctor says the generic is fine. Why? Because of misinformation. A survey found that 43% of people believe generics contain only 20-80% of the active ingredient. That’s false. The FDA’s own testing shows they contain 99% or more.
What’s changing now?
The system is getting faster. The FDA’s GDUFA IV program, running from 2023 to 2027, is investing $1.12 billion to cut approval times for generics from 38 months down to 10 months by 2027. Why? Because more drugs are coming off patent. In 2023 alone, $28 billion in brand-name sales became eligible for generic versions-including big names like Eliquis and Entyvio.
Biosimilars-generic versions of complex biologic drugs-are also growing fast. While they’re not identical to the original (because biologics are made from living cells), they’re held to the same high standards. By 2027, they’re expected to make up 15% of the market.
The real win? Cost savings. The 2022 Consolidated Appropriations Act now requires Medicare Part D to automatically switch to generics unless there’s a medical reason not to. That’s projected to save $156 billion over ten years.
Bottom line: Generics work. They’re safe. And they save lives.
Generic drugs aren’t copies in the way a pirated movie is a copy. They’re scientifically identical, legally required to be identical, and tested to prove it. The differences you see-color, shape, price-are superficial. The medicine? It’s the same.
For the vast majority of people, switching to a generic is the smartest health decision you can make. It’s not about saving money-it’s about staying on your medication. Studies show that people who can’t afford brand-name drugs are 25% more likely to skip doses. With generics, that drops to 8%.
If your doctor says a generic is fine, trust it. If you feel something’s off, talk to your pharmacist. But don’t assume the worst because it looks different. The science is clear: generics work.
Are generic drugs less effective than brand-name drugs?
No. Generic drugs must meet the same FDA standards as brand-name drugs. They contain the same active ingredient, in the same strength, and work the same way in your body. The FDA requires bioequivalence testing to prove this. Over 90% of prescriptions in the U.S. are for generics, and studies show they perform just as well in real-world use.
Why do generic drugs look different?
Federal law requires generic drugs to look different from brand-name versions to avoid trademark infringement. This means the shape, color, or logo might change, but the active ingredient is identical. Differences in appearance have no effect on how the drug works.
Can I switch from a brand-name drug to a generic safely?
Yes, for most medications. Over 96% of non-NTI (narrow therapeutic index) drugs, like statins, blood pressure pills, and antidepressants, can be safely switched. For NTI drugs like warfarin or levothyroxine, your doctor may monitor you more closely, but switching is still common and safe for most patients.
Why are generic drugs so much cheaper?
Brand-name drugs cost more because the manufacturer paid for research, clinical trials, and marketing. Generic manufacturers don’t repeat those steps-they only prove their version works the same. This cuts costs dramatically. On average, generics cost 85% less than brand-name drugs.
Do pharmacists get paid more to give me the brand-name drug?
No. Pharmacists don’t earn more from prescribing brand-name drugs. In fact, many pharmacies make more profit on generics because they’re cheaper to buy and easier to stock. Pharmacists are trained to recommend generics when appropriate-it’s part of their job to help you save money without sacrificing care.
What if I think the generic isn’t working for me?
Talk to your pharmacist or doctor. Sometimes side effects come from inactive ingredients (like dyes or fillers), not the active drug. If you’re on a critical medication like an antiseizure drug or thyroid medicine, your provider may want to check your blood levels. But in most cases, switching back to the brand isn’t necessary-just a different generic brand might help.
If you’re unsure, ask. But don’t let myths keep you from the most affordable, proven option out there. Your body doesn’t care what the pill looks like. It only cares if the medicine works-and for generics, it always does.






Dylan Patrick
March 17, 2026 AT 10:05Generics saved my life. I was on Lipitor for years, paying $200 a month. Switched to generic atorvastatin? $4. Same pill, same results. No weird side effects. My doctor said it’s identical, and honestly? My cholesterol’s never been better. Stop overthinking the color.
People act like generics are some shady pharmacy scam. Nah. They’re just capitalism working for once.
Kathy Leslie
March 18, 2026 AT 01:41I used to freak out every time my pill changed shape. Then I started reading the FDA stuff. Turns out, it’s literally the same chemical. I even called my pharmacist. She laughed and said, ‘It’s like buying a different brand of aspirin.’
Now I don’t even look at the pill. Just take it. Life’s too short to stress over dye.
Leah Dobbin
March 19, 2026 AT 04:31It’s fascinating how the general public conflates appearance with efficacy. The FDA’s bioequivalence standards are not merely rigorous-they are mathematically stringent, with a 90% confidence interval anchored between 80% and 125% AUC and Cmax. To suggest that a pill’s hue or logo implies inferiority is to misunderstand pharmacokinetics at a fundamental level.
One might as well argue that a BMW and a Toyota Corolla are mechanically different because one has a chrome trim.
Stephanie Paluch
March 19, 2026 AT 07:53My mom switched to generic levothyroxine and started feeling dizzy. We panicked. Then we found out the new batch had lactose. She’s lactose intolerant. Switched to a different generic without lactose? Back to normal.
It’s not the drug. It’s the filler. Always ask your pharmacist about inactive ingredients. 💬❤️
Serena Petrie
March 20, 2026 AT 23:40My pill changed color. Now I feel weird. Probably the generic. 🤷♀️
Elsa Rodriguez
March 22, 2026 AT 13:49OMG I switched to generic metformin and my stomach went full apocalypse. I thought I was dying. Turns out it had a different coating. My pharmacist said ‘it’s the same drug’ but I swear I felt it in my soul.
Why do they even make us take this risk?? I’m never switching again. #GenericTrauma
tynece roberts
March 22, 2026 AT 20:07so like i was on brand name sertraline for 3 years and then switched and at first i thought it wasnt working but then i realized i was just stressed out and my brain was overthinking it. like the pill looks different so my brain went ‘this is not the same thing’ but it is. its literally the same molecule. i even checked the label. same dosage. same everything. just a different shape. why do we make this so hard?
also my pharmacist gave me a free sample of a different generic and it worked better. idk why. maybe the binder? idk. but i stopped crying about it.
Aaron Leib
March 23, 2026 AT 23:08For 96% of meds, generics are perfect. The FDA tests every batch. The math doesn’t lie.
Worried about your thyroid or seizure med? Talk to your doc. But don’t assume the worst. Most people who think generics don’t work are reacting to fillers, not the drug.
Pharmacists aren’t pushing generics to make money. They’re pushing them because they work. And they save you hundreds.
Rosemary Chude-Sokei
March 25, 2026 AT 16:58As a healthcare professional with over two decades of clinical experience, I have observed that the perception of inferiority surrounding generic medications is not grounded in empirical evidence, but rather in sociocultural narratives perpetuated by pharmaceutical marketing. The FDA’s bioequivalence requirements are among the most stringent in the world, and the regulatory oversight is exhaustive.
Moreover, the economic impact of generic adoption cannot be overstated. In low-income populations, the difference between adherence and non-adherence is often the difference between life and death. Generics are not a compromise-they are an ethical imperative.
It is imperative that clinicians and patients alike reject misinformation and embrace evidence-based decision-making. The data is unequivocal: generics are therapeutically equivalent, and their widespread use is a public health triumph.
mir yasir
March 27, 2026 AT 11:54One must question the validity of the FDA’s bioequivalence thresholds. A confidence interval of 80–125% is statistically broad, especially when considering drugs with narrow therapeutic windows. While the aggregate data may appear favorable, individual pharmacogenomic variance is rarely accounted for in these studies.
Furthermore, the global harmonization of standards is misleading-regulatory agencies in developing nations lack the infrastructure for independent batch verification. The assumption that approval in one jurisdiction guarantees equivalence elsewhere is a dangerous oversimplification.
Noluthando Devour Mamabolo
March 27, 2026 AT 13:40Bro, I switched to generic warfarin and my INR went haywire. My doc said ‘it’s bioequivalent’ but my blood was all over the place. I switched back to brand. Now I’m stable. So yeah, for NTI drugs? Not all generics are created equal. 🤷♂️💊
Also, the FDA’s 92% success rate? That’s still 8% of people who got messed up. That’s not ‘rare’-that’s 1 in 12. I’m not a statistic.
Dylan Patrick
March 28, 2026 AT 13:45Actually, that’s why NTI drugs are monitored. Your doc should’ve checked your INR 2 weeks after switching. Most people don’t. That’s the problem-not the drug.
I switched my mom’s levothyroxine three times. Each time, her TSH was stable within 0.5 points. The only difference? She stopped having heartburn. Turns out, the brand had a dye she was allergic to. Generic? No dye. No issues.